Introduction
Co-expression of C-MYC and BCL2 in diffuse large b-cell lymphomas (DLBCL), also termed double-expressor lymphomas (DEL), has been shown to be associated with poorer outcomes after standard R-CHOP induction therapy. Approximately 70% of DEL will relapse within 5 years. The subsequent traditional approach with potential for cure has been high dose chemotherapy followed by autologous hematopoietic stem cell transplantation (ASCT). Whilst factors such as remission status after front-line treatment and early relapse have been associated with poorer outcomes after ASCT, data are limited regarding significance of DEL status on survival outcomes post-ASCT. We retrospectively studied the prognostic impact of DEL status on outcomes in patients with relapsed or refractory (R/R) DLBCL who underwent ASCT.
Methods
This was a retrospective, nationwide study of adults patients, >18 years old, with R/R DLBCL who underwent autologous hematopoietic stem cell transplantation (ASCT) between 2010 and 2022 at the only 2 transplant centers in Singapore - Singapore General Hospital and National University Cancer Institute. Patients with double/triple hit lymphoma, defined as concurrent rearrangements of MYC and BCL2 and/or BCL6 by fluorescence in situ hybridization (FISH) analysis, were not included in this study. Patients with transformed indolent B-cell NHL (non hodgkin lymphoma), primary mediastinal B-cell lymphoma, primary CNS (central nervous system) lymphoma, or Richter transformation of chronic lymphocytic leukemia were excluded. Only patients that had available MYC and BCL-2 immunohistochemistry (IHC) were included in this study. Overall survival (OS) and progression free survival (PFS) were estimated using the Kaplan-Meier method. Cox proportional hazards regression models were used to assess associations between covariates of interest and OS or PFS.
Results
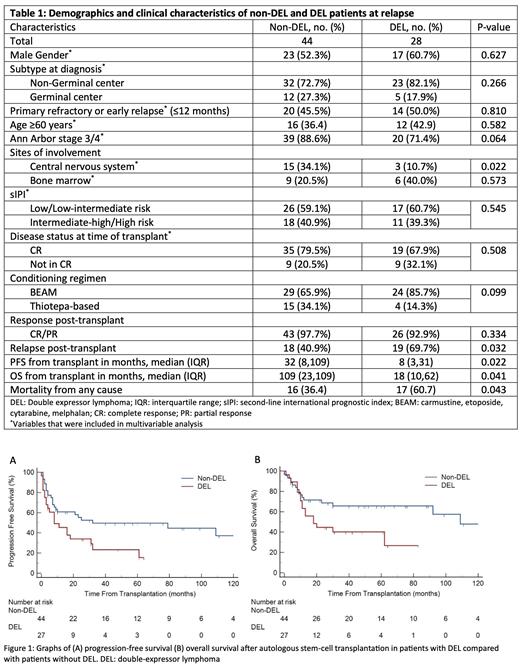
A total of 72 patients were included, 44 (61.1%) non-double expressor DLBCL (non-DEL) and 28 (38.9%) double expressor DLBCL (DEL). Patient demographics and disease characteristics at relapse were similar in both groups aside from a higher incidence of CNS involvement in the non-DEL group as compared to DEL group, 34.1% vs 10.7% (Table 1). Both groups achieved a >90% response (CR and PR) after second-line salvage chemotherapy as well as a >90% response post-transplant (CR and PR). Patients with DEL however were more likely to relapse post-transplant, 69.7% vs 40.9% (p = 0.032) and were also associated with increased odds of all-cause mortality, 60.7% vs 36.4% (p=0.043). This significance was maintained on multivariable analysis, HR 4.22 95% CI 1.20 - 14.84 (p=0.025) and HR 4.32 95% CI 1.10 - 17.15 (p=0.038) respectively. The median follow up for survivors were 42 months (range, 26 to 64 months) in the DEL patients and 60 months (range, 46 to 88 months) in the non-DEL patients. The 5-year PFS in patients with DEL was inferior to non-DEL patients, 23.3% vs 49.6% respectively, p=0.022. DEL patients were also associated with inferior 5-year OS, 40.2% vs 65.7%, p=0.041. In multivariable model, DEL status remained significantly associated with inferior PFS and OS. We also observed that patients with CNS involvement at relapse had inferior PFS and OS. Male gender and a less than complete metabolic response (CR) status prior to transplant, assessed on PET-CT, was also associated with inferior OS.
Conclusion
Co-expression of C-MYC/BCL2 was associated with inferior outcomes after ASCT in patients with R/R DLBCL. While ASCT has been the standard curative approach in the R/R setting, considerations for novel targeting therapies to achieve a deeper response prior to transplant or the upfront use of chimeric antigen receptor T-cell therapy at first relapse may be further explored in this high-risk subset of patients.
Disclosures
Nagarajan:BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees; DKSH/Beigene: Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: The Trial was supported by funding to IMF/AMN who were the sponsorswere the Sponsors; Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees; Astrazeneca: Honoraria, Membership on an entity's Board of Directors or advisory committees.